GV104 represents a novel advancement in pharmaceutical innovation, serving as a cornerstone for our lead clinical program. This product is meticulously crafted by embedding a drug onto a proprietary transmucosal buccal film, designed to adhere seamlessly to the inner lining of the mouth. This unique formulation of GV104 positions it as an optimal candidate for addressing a spectrum of conditions, with our lead program addressing a critical need to protect military personnel and chemical incident responders.
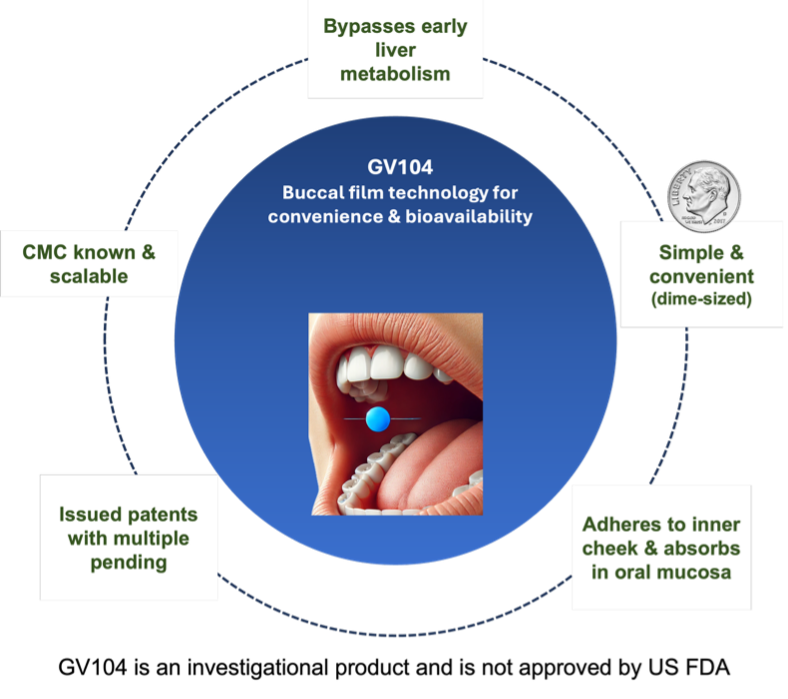
Gravitas Life Sciences, Inc. is seeking to develop GV104 for the proposed indication of “Temporary Prophylaxis of Respiratory and/or Nervous System Depression in Military Personnel and Chemical Incident Responders Entering and Area Contaminated with High-Potency Opioids”. Gravitas Life Sciences recently received positive feedback from the U.S. Food and Drug Administration (FDA) regarding a regulatory pathway that will allow the Company to submit a 505(b)(2) New Drug Application (NDA) for GV104.
The FDA has confirmed that no additional clinical trials will berequired prior to NDA submission for this indication.
The foundation for the FDA’s alignment on the submission of an NDA without additional clinical trials rests on the extensive existing data related to the approved active ingredient nalmefene embedded in GV104. This includes a comprehensive understanding of its safety profile, established through years of use in approved products, and evidence of its effectiveness against certain opioid receptors.
Our strategic roadmap for GV104 began with completion of our phase 1 trial and phase 2 readiness to study patients with a specific type of debilitating itch in a rare liver condition. Leveraging our positive feedback from FDA and EMA, we anticipate further development of GV104 in commercial applications following a more near-term NDA filing strategy for prophylaxis against ultrapotent opioid exposure.
Gravitas Life Sciences
powered by Tharimmune
Next Generation of Oral Infliximab

*Phase 2 Clinical CMC completed; HI – hepatic impairment; GV104 is Phase 2 ready in the EU and US with FDA and EMA feedback received
**trial initiation ex-US; Celltrion has right-of-first refusal post Phase 2
†Gravitas Life Sciences received positive feedback from the FDA regarding a regulatory pathway that will allow the Company to submit a 505(b)(2) New Drug Application (NDA) for GV104. Importantly, the FDA has confirmed that no additional clinical trials will be required prior to NDA submission for this indication.
Lead Program: GV104
With the completion of a Phase 1 trial, we engaged and received positive feedback from the FDA regarding the additional proposed indication of temporary prophylaxis of respiratory and/or nervous system depression in military personnel and chemical incident responders entering and area contaminated with high-potency opioids. The Company received positive feedback from the FDA regarding a regulatory pathway that will allow Gravitas Life Sciences to submit a 505(b)(2) New Drug Application (NDA) for GV104. The FDA confirmed that no additional clinical trials will be required prior to NDA submission for this indication, which is the most advanced program for the Company.

